Prioritize Your Proteins
Note to readers: You can listen to this article by clicking on the voiceover recording above.
“Proteins are the compounds that define most of the properties we ascribe to life.” 1
Our early ancestors were hunters and foragers, men and women who lived in tune with the earth in ways we can scarcely imagine today. Both in and of the ecosystem that sustained them, they were inseparable from the context that gave rise to them. Their flesh and bones were woven of the elements of the mountains, valleys, and forests where they lived. Their prehistoric partnership with these elements was knitted into the root level of their cells. Billions of biochemical pathways evolved to sustain their long-ago lives. The plants, animals, fungi, and microorganisms they consumed co-evolved along with them, creating an ever more intricate web of interdependence. Nature’s design is inconceivably old, and truly awe-inspiring.
Our bodies are still designed and programmed according to this ancient blueprint. In terms of our cellular functioning, we haven’t evolved much at all – though in recent generations our jaws have shrunk from eating low-fiber foods, and along with that, our sinus cavities. With the rise of antibiotics and many other gut-busting drugs, our microbiomes have ceased to pass down the beneficial gut bacteria that kept our great-grandparents healthy. Yet despite our efforts to sabotage nature’s design, we are still beholden to it. When we harmonize with this design, our bodies (including our brains) are metabolically and structurally optimized. When we don’t, we necessarily break down.
Food in its whole form may be regarded as a consortium of nutrients – a symphony of proteins, fats, carbohydrates, micronutrients (minerals, vitamins, phytochemicals), and fiber. All are essential for human health. This article focuses on protein, the building blocks of our bodies. The foods we eat contain vastly different combinations of proteins that are broken down to become our tissues and fluids, and much of our immune system. Amino acids, the basic components of proteins, may also act as chemical messengers, such as to transmit impulses between nerves, or as carriers of minerals that enable enzymes to work. Proteins make up the most essential parts of our bodies: from hormones to hemoglobin, from muscles to membranes, from nerve cells to neurotransmitters – and more. An adult human contains thousands upon thousands of different proteins that constellate and reconstellate within us every day, like the stars in heaven dancing and wheeling through time.
“Proteins determine our metabolism, form our tissues, give us motion, transport compounds, and protect us from deleterious invasion. Even the heredity of an organism is nothing more than an expression of its ability to make various kinds of proteins at different rates.” 2
How Proteins Work
The structure of proteins serves their many roles in our bodies. They are synthesized in the nucleus of our cells from 20 different amino acids. Free amino acids absorbed from protein foods into the cytoplasm of cells provide the raw materials for building short chain peptides as well as more complex polypeptides. Several organelles within the cell oversee the final formation and editing of these structures.
Thus, eating protein supplies your body with what it needs to create proteins endogenously. Collagen in your bone broth is not directly integrated into your joints; rather, it is broken down into proline and glycine, amino acids which are then used to synthesize collagen.
Like all molecules, amino acids consist of atoms that may be either positively or negatively charged. Opposites attract, shaping both the form and behavior of chemical bonds, which in turn trigger different reactions. Protein molecules also have the capacity to be neutral, or both positive and negative at the same time – known as a zwitterion. And they are sensitive to acid and basic environments, in which they may become more or less stable and soluble.
These shape-shifting capacities are the genius of proteins. Like microscopic origami, they literally fold into different configurations – all manner of helixes, rods, envelopes, and other structures ranging from simple to elaborate. If misfolding or tangling damages this structure, the protein cannot play its part.
An example of this is glycation, when proteins and sugars melt and fuse together in foods cooked at high temperatures, resulting in smoked, roasted, fried or crispy textures. Toasted bagels and crispy French fries are two examples of glycation. Glycated proteins are irreparably damaged, and when consumed in foods, do not perform well internally. The result is most visible in malformed collagen fibers, resulting in wrinkled skin and connective tissue problems such as trigger finger. It is not aging per se that causes wrinkles, but the cumulative effect over time of diminishing nutrient absorption, declining protein synthesis, and damaged collagen fibers.
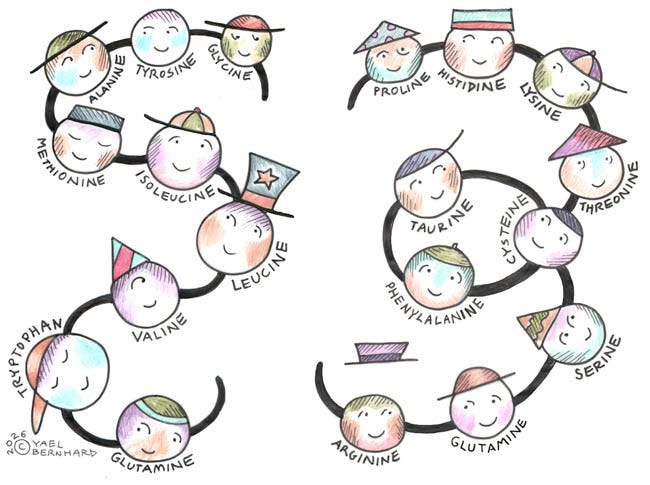
All amino acids are composed of a core structure consisting of two atomic groups: a nitrogen-based amino group and a carbon-based carboxyl group. In between is where the difference lies: the “side chain” attached to this core determines the characteristics of each amino acid. Each has a role to play – or several, for proteins are pleomorphic, with the ability to change form and function. These little fellows wear many different hats!
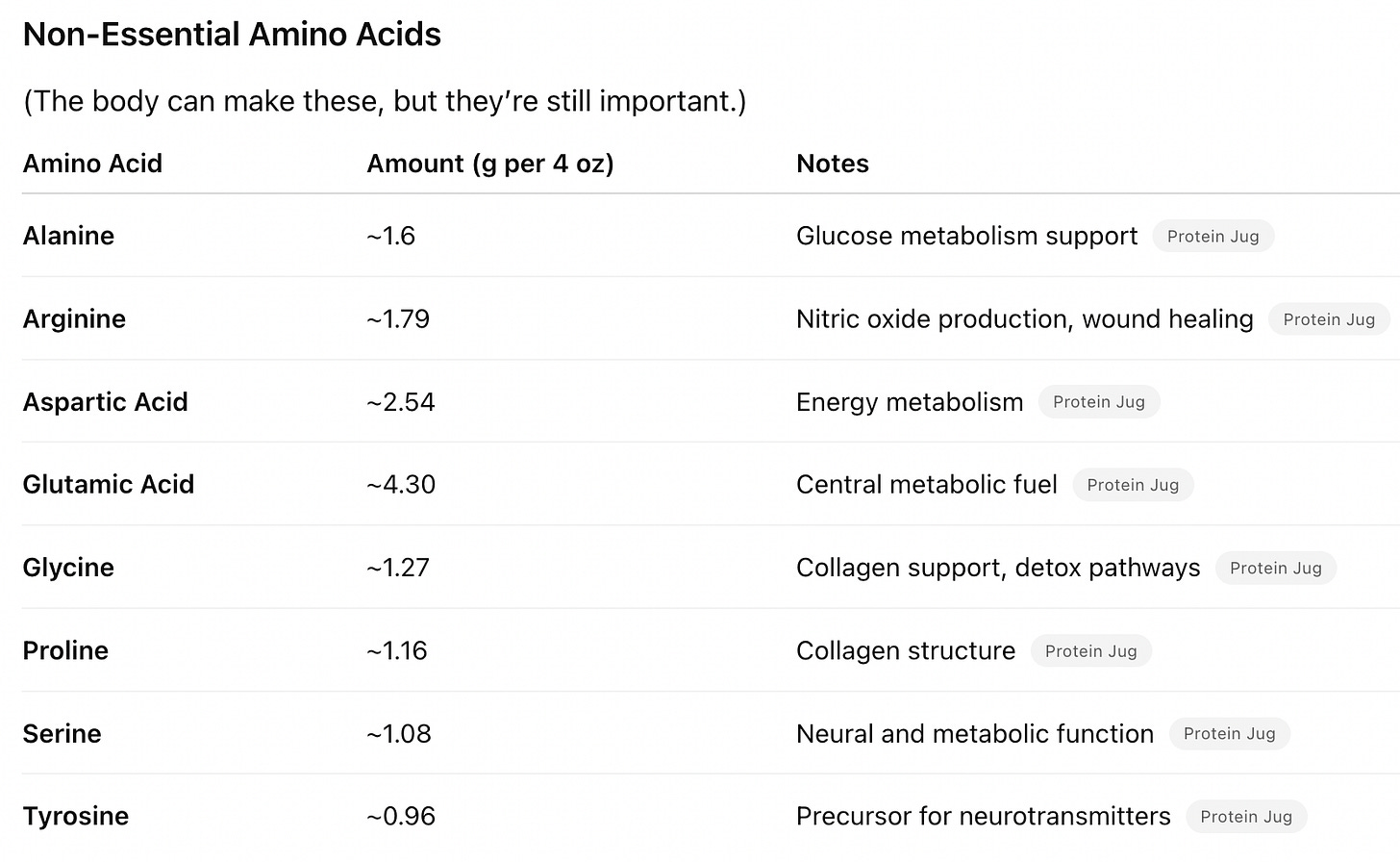
Amino acids are classified as “essential” (necessary to derive from one’s diet) or “non-essential” (produced endogenously, not made directly from food). Some amino acids work together, and must be available in the right proportions. Not only is it critical to eat enough protein, but also to get the right proportion of amino acids in the foods you eat. Let’s take a look at some of these hidden partnerships:
Lysine and glutamine work together to stabilize enzyme activity, enabling millions of enzymatic reactions to work properly.
Methionine supplies methyl groups for DNA methylation and growth, while glycine prevents the excess cell division of cancer. Excess methionine may also result in elevated homocysteine, a marker of cardiovascular disease. Methionine is derived from muscle meat, while glycine comes from organs, skins, bones, and joints. For hundreds of thousands of years, our ancestors ate the animals they hunted “from nose to tail,” ensuring a proper balance between methionine and glycine. Most modern-day omnivores tend to eat only muscle meat, thus risking overactivity of cellular methylation. Consuming bone broth or collagen, which contain glycine, corrects the imbalance.
Phenylalanine converts into tyrosine, which then makes dopamine, norepinephrine, and thyroid hormones – all essential for human health.
Excitatory glutamate pairs with GABA, the calming neurotransmitter; while tryptophan and tyrosine balance each other in transporting serotonin, melatonin, dopamine, and norepinephrine for proper uptake.
Proline and glycine are necessary to form collagen, the most prevalent protein in your body and the basis of all structures including bones, joints, hair, skin, nails, connective tissue, membranes, and more.
Proline and lysine work together to form the cross-linking that completes and strengthens collagen.
Glutamine and alanine transport nitrogen to the liver, and maintain stable blood sugar during fasting.
Cysteine pairs with itself to form sulfur-based bridges that stabilize the 3-dimensional structures and foldings in proteins, and supports enzyme reactions. Together with glycine and glutamate, cysteine forms your “master antioxidant,” glutathione, especially important for liver function and detoxification. Alcohol and Tylenol directly deplete cysteine. Whey protein is an excellent source.
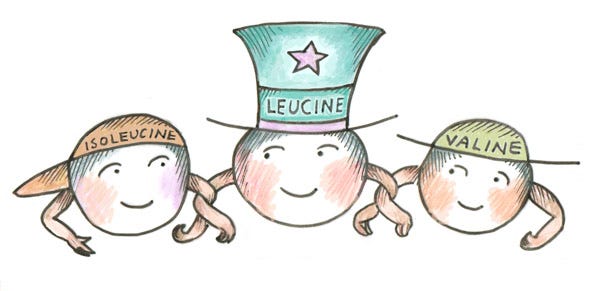
Leucine and isoleucine pair up to stabilize the interior structure of complex proteins. They are hydrophobic, resisting water to keep the protein stable.
Leucine is a rate-limiting amino acid: protein synthesis is limited to the amount of leucine present. Leucine turns on protein synthesis, something to keep in mind when choosing your protein foods. In addition, leucine must be balanced with valine and isoleucine in order to function properly. These are known as branch-chain amino acids (BCAAs) because of the atomic structure of their branched side chains.
With the exception of soybeans (which contain 2.5g of leucine per cup of cooked beans), leucine is present only in modest or trace amounts in plants. Relying heavily on soy for adequate leucine, however, is fraught with problems, such as the use of harmful weedkillers and dessicants in mass agriculture (see my article Eating for the Environment for more on this subject) and the presence of phytoestrogens in this plant. Fermented soy, such as miso, tamari, and tempeh, is generally healthier, depending on the state of your microbiome. But by far, animal protein is a superior source of BCAAs, and presents a perfect package of balanced amino acids. This is no accident – it’s nature’s design, and what our bodies are evolved to thrive on.
How much protein do we need?
New federal guidelines just published this month increase the recommended amount of protein from 0.8 grams per kilogram of body weight to 1.2-1.5 grams (or roughly .75g per pound of body weight). This is a welcome step in the right direction, but it is contradicted by the continuing cap on saturated fat.3 Recommendations for protein vary by age and sex. Growing children, older adults, pregnant and nursing women, and very active individuals need more. Sedentary people over the age of 40 also need more protein, because without exercise, we cannot enter an anabolic (protein-building) state, and protein synthesis diminishes.
More progressive, functional models echo this new call for more protein – and more exercise – making a clear distinction between “adequate” levels (to prevent deficiency) and “optimal” amounts (for metabolic health, strong immunity, and best possible longevity). As an active, aging adult who exercises daily, I shoot for 70-90g/day, or roughly 1.5g/kg of my body weight.
It’s worth keeping a 3-day protein diary to determine if you’re getting enough. You might be surprised! Some people eat more protein than they realize, while others are consuming far less. AI makes it easy to find out what’s in the specific portions you eat.
What is a complete protein?
In 1971, Diet for a Small Planet introduced the concept of protein-combining to the American public. The book sparked a revolution of plant-based diets, yet later editions retracted its initial assertions, replacing strict combining with a broader approach. Still, its main thesis asserted that plant-based diets may easily replace meat.
Does this theory still hold true over fifty years later? Partially, perhaps. Along with half a century of plant-based diets have come rising rates of diabetes, obesity, cardiovascular disease, and cancer; as well as persistent signs of protein inadequacy, especially among vegans and aging adults. These include: muscle wasting; low energy; anemia; slower recovery after exercise; feeling drained even after sleep; thinning or brittle hair; sugar cravings; increased appetite; weak nails; poor wound healing; frequent colds or infections; brain fog; low mood; swollen ankles or feet; and low bone density. Other factors besides choice of diet may contribute to these symptoms, such as the rise of ultra-processed foods (which are highly inflammatory) and factory-farmed meats (which lack the micronutrients necessary for protein assimilation due to the animals’ poor diets and health). But the trend is still evident – and it’s no mystery, for animal proteins are more easily digested and assimilated by our bodies than plant proteins, which are locked behind cellulose walls rather than phospholipid cell membranes. We are made of animal proteins, and we’re designed to spin our bones, blood, skin and flesh from the proteins of other animals.
This is especially crucial as we age. When we’re young, protein synthesis is largely driven by hormones; but as we get older, it slows down, and is only stimulated by exercise. Loss of muscle mass (sarcopenia) is a major contributor to aging, and underlies many life-limiting injuries. Along with the diminishing levels of protein that drive sarcopenia comes lower immune response, for this too depends on protein. To limit one’s ability to synthesize protein by eating the wrong proportions only adds to the problem. Total protein is not all that matters! The proportions of your amino acids matter very much, and as we have seen, so do their combinations.
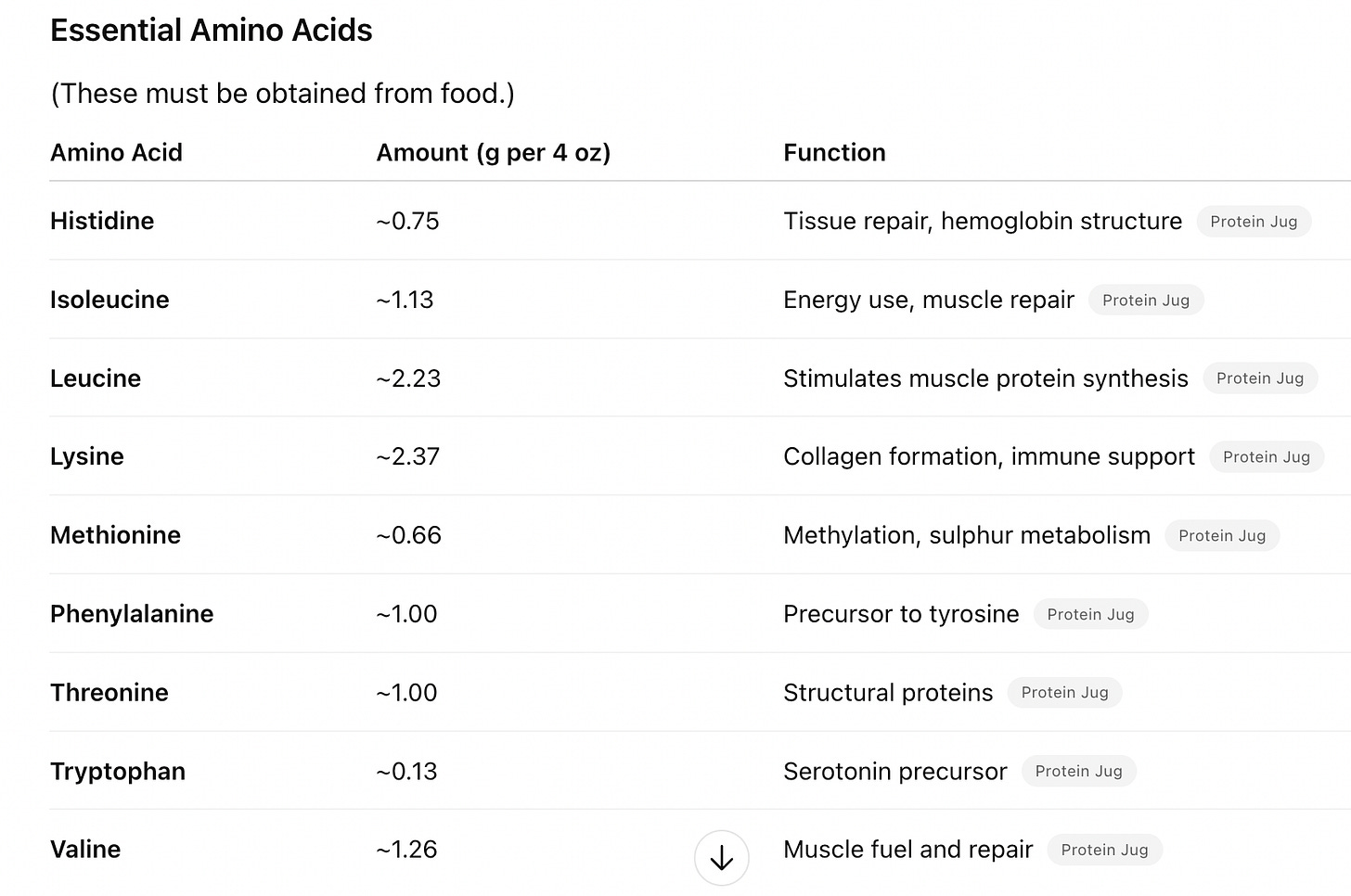
To shed some light on this issue, let’s examine the amino acid profile of a 4oz serving of grass-fed beef, containing 20g of total protein:
Note: Cysteine is measured separately and would come to .3-.4g in this context. It is more abundant in the joints, hides, and bones of animals than in muscle meat.
Taurine is also not included here because it is a “conditional” amino acid that is different in its structure. Taurine is necessary for many vital bodily functions, such as cell stabilization, nerve impulses, and antioxidant activity. It's found naturally in meat, fish, and dairy, and is synthesized in the body. A 4oz serving of grassfed beef would contain .4-.6g of Taurine.
Beef is a complete source of essential amino acids — all nine are present at meaningful levels. Those all-important BCAAs are especially abundant, and they are balanced with their amino acid partners. If taken from a healthy animal, beef is an excellent source of both complete protein and heme iron (which binds oxygen to the proteins in hemoglobin, to be delivered to every cell in your body for cellular respiration). Grass-fed beef is superior, containing healthier fats and micronutrients.
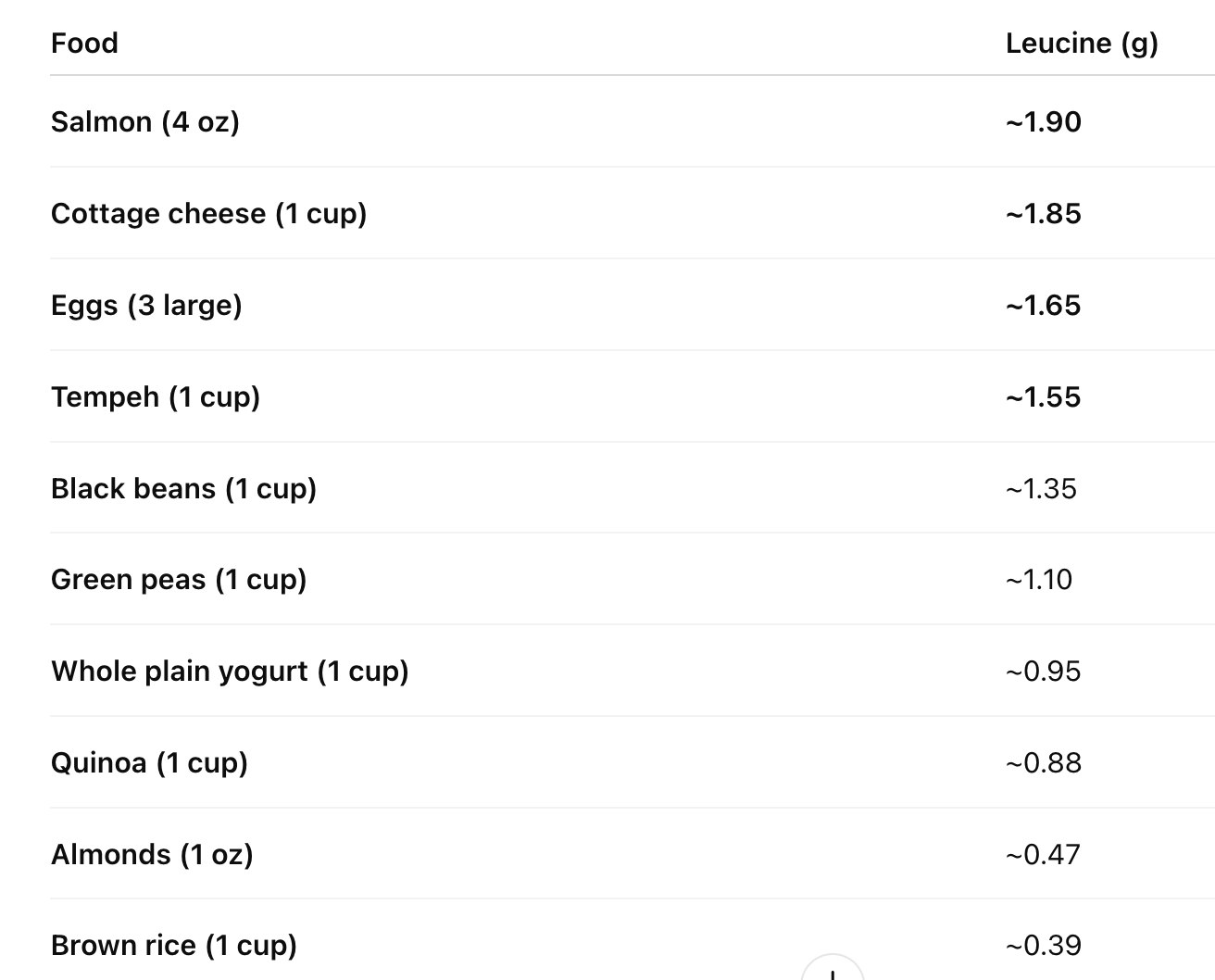
Now let’s look at the leucine content of some other common protein foods:
From this information, we have several key takeaways:
A complete protein has all nine essential amino acids in sufficient proportions. Animal proteins (meat, fish, eggs, cheese, yogurt) deliver much more BCAAs per calorie and per gram than plant proteins.
Due to pairing activity, whole proteins behave differently than isolated amino acids.
~2–3 g leucine per meal is often cited as a rough threshold for maximal muscle protein synthesis in adults. This is easily reached with animal protein, and requires larger and combined portions of plant foods.
Legumes (beans, peas) are good BCAA sources, but come with excessive starchy carbohydrates. Many more calories – 4-6 times as much – must be consumed to reach adequate levels. Unless carefully monitored and combined with regular exercise, a diet of exclusively plant-based proteins may result in diabetes, obesity, or both.
Grains (rice, quinoa) contribute modestly and usually need pairing with other proteins.
Why Whole Proteins?
Animal protein is the most nutrient-dense food on the planet. Along with it comes other micronutrients that are necessary for human health and longevity: for example choline, vitamin D, vitamin B12, retinal (the form of vitamin A necessary for healthy vision), and heme iron. These may only be derived from animals. Other nutrients are far more abundant and usable from animal sources, such as zinc, omega-3 essential fatty acids, and creatine. Pasture-raised meat is also rich in healthy saturated fats, and in the phytonutrients contained in the wide array of plants consumed by grazing ruminants. The prefix “phyto” refers to plants, but animals also consume fungi, insects, soil, and microorganisms. Through these animals, we are intricately connected with the entire soil-food web.
It’s not possible to pick these elements apart and deliver them in conveniently-packaged products. Like all foods, protein is best consumed in its whole form. If it is not possible to make it yourself, not recognizable as food, and you cannot detect the true ingredients with your own senses, it’s not a whole food. Processed, powdered, artificially-flavored, and packed-in-plastic protein is not whole. Plant protein isolates may also concentrate unwanted ingredients, such as pesticides and heavy metals.4 I’m not here to dissuade you from using protein powders and drinks, but it is not realistic or healthy to rely on them for sufficient, complete protein.
Finding the right source
As for quality and type of animal proteins we choose to eat, that is the subject of a forthcoming article. Quality and source matter. Animal welfare matters. Impact on the ecosystem matters. This is a controversial subject that touches upon environmental, ethical, economic, and other considerations. Your choices begin with an understanding of protein, why you need it, and the many roles it plays in your body.
Clearly, not all protein is created equal, and it takes more than a bowl of beans and rice to get the right proportions. After all, rice is native to Asia, and beans to the Americas. They were first combined during the colonial era, only about 500 years ago with the European conquest of Central America. In terms of human evolution, that is not even a scratch in time. Our bodies aren’t designed to live on beans and rice, just as a banana tree can’t thrive in Kansas. It might survive for a while, but in the long run it cannot overcome its intrinsic tropical nature.
My fascination with proteins has led me down deep rabbit holes that have opened into wildly branching warrens. The result is a diet of moderate meat intake (several servings of wild-caught fish and grass-fed meat per week), with other sources of animal protein such as eggs, yogurt, and cheese consumed daily. After ten years of vegetarianism, twenty years of hunting my own meat, and a lifetime of cooking, gardening, foraging, butchering, and learning about nutrition, I’m firmly convinced that we may only build and sustain long-term health by cooperating with nature’s design. The question is not whether or not we’re omnivores, but how.
My goal is to harmonize with the environment I inhabit, and the earth as a whole. All around me are its atmospheric forces, its great and flowing waters, its shifting sands and crumbling rocks. What an incredible design, and what a privilege to be part of it, and to walk in the biological footsteps of my ancestors! Whether a microscopic molecule, a network of soil organisms, a herd of animals or a constellation of stars, it’s all part of the same immense and awe-inspiring web of life.
To your good health, and your place in that vast and intricate web –
Yael Bernhard
Certified Integrative Health & Nutrition Coach
Yael Bernhard is a writer, illustrator, book designer and fine art painter with a lifelong passion for nutrition and herbal medicine. She was certified by Duke University as an Integrative Health Coach in 2021 and by Cornell University in Nutrition & Healthy Living in 2022. For information about private health coaching or nutrition programs for schools, please respond directly to this newsletter, or email dyaelbernhard@protonmail.com. Visit her online gallery of illustration, fine art, and children’s books here.
Information in this newsletter is provided for educational – and inspirational – purposes only.
Have you seen my other Substack, Image of the Week? Check it out here, and learn about my illustrations and fine art paintings, and the stories and creative process behind them.
McGilvery, Robert PhD and Goldstein, Gerald MD, Biochemistry: A Functional Approach (college textbook), W.B. Saunders Publishing, 1983, pg. 3.
Ibid.
The 10% cap means approximately 20-22 grams of saturated fat daily for the average 2,000-calorie diet. It is nearly impossible to meet the recommended protein intake without exceeding this limit (for example, an egg, which is an excellent source of protein and other vital nutrients, contains 16% saturated fat). It is my opinion that saturated fat from healthy animals, if eaten in the context of a healthy lifestyle (including regular exercise), is safe beyond the recommended limit. Despite media buzz warning the public about saturated fat, there is virtually no research linking it to heart disease, cancer, or any other cause of mortality. These warnings are politically-motivated.
Apple podcast: what-you-need-to-know-about-lead-and-other-heavy/id1381257272?i=1000736409595




Thank you for your comment!